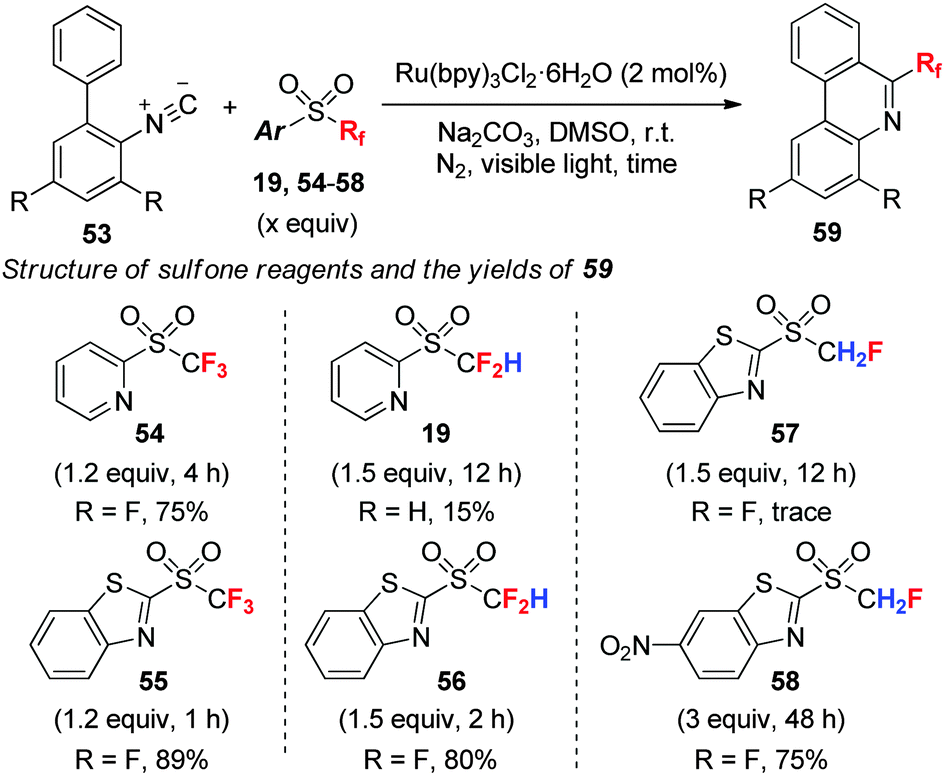
Upon cooling fluorine becomes a yellow liquid. Physical and chemical propertiesĪt room temperature fluorine is a faintly yellow gas with an irritating odour. The principal fluorine-containing minerals are (1) fluorspar, deposits of which occur in Illinois, Kentucky, Derbyshire, southern Germany, the south of France, and Russia and the chief source of fluorine, (2) cryolite (Na 3AlF 6), chiefly from Greenland, (3) fluoroapatite (Ca 5 3), widely distributed and containing variable amounts of fluorine and chlorine, (4) topaz (Al 2SiO 4 2), the gemstone, and (5) lepidolite, a mica as well as a component of animal bones and teeth. Not a rare element, it makes up about 0.065 percent of Earth’s crust.
Fluorine reactivity free#
The colourless, transparent crystals of fluorspar exhibit a bluish tinge when illuminated, and this property is accordingly known as fluorescence.įluorine is found in nature only in the form of its chemical compounds, except for trace amounts of the free element in fluorspar that has been subjected to radiation from radium.

The name fluorspar is derived from the Latin fluere, “to flow.” The mineral subsequently proved to be a source of the element, which was accordingly named fluorine. The fluorine-containing mineral fluorspar ( fluorite, CaF 2) has been used for centuries as a flux (cleansing agent) in various metallurgical processes. Then, however, the use of uranium hexafluoride in the separation of uranium isotopes, along with the development of organic fluorine compounds of industrial importance, made fluorine an industrial chemical of considerable use. Indeed, up to the time of World War II the element appeared to be a laboratory curiosity.
The difficulty in handling the element and its toxic properties contributed to the slow progress in fluorine chemistry. He received the 1906 Nobel Prize for Chemistry for isolating fluorine. The isolation of fluorine was for a long time one of the chief unsolved problems in inorganic chemistry, and it was not until 1886 that the French chemist Henri Moissan prepared the element by electrolyzing a solution of potassium hydrogen fluoride in hydrogen fluoride. How well do you know their symbols? In this quiz you’ll be shown all 118 chemical symbols, and you’ll need to choose the name of the chemical element that each one represents. The periodic table is made up of 118 elements. Fluorspar was then recognized to be calcium fluoride. The nearly anhydrous acid was prepared in 1809, and two years later the French physicist André-Marie Ampère suggested that it was a compound of hydrogen with an unknown element, analogous to chlorine, for which he suggested the name fluorine. In 1771 the Swedish chemist Carl Wilhelm Scheele obtained hydrofluoric acid in an impure state by heating fluorspar with concentrated sulfuric acid in a glass retort, which was greatly corroded by the product as a result, vessels made of metal were used in subsequent experiments with the substance. It appears likely that crude hydrofluoric acid was first prepared by an unknown English glassworker in 1720. The fluorine-containing mineral fluorspar (or fluorite) was described in 1529 by the German physician and mineralogist Georgius Agricola. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.
Fluorine reactivity how to#

From tech to household and wellness products.
